 
To find the molar mass of SO3, you would first need to find the atomic mass of each element. 0:000:47Molar Mass / Molecular Weight of SO3 : Sulfur dioxide – YouTubeYouTubeStart of suggested. Sulfur trioxide | SO3 or O3S | CID 24682 – structure, chemical names, physical and chemical properties, classification, patents, literature, What is the molecular weight of sulfur trioxide?. , shows that its molecules contain one S atom and two O atoms calculate its molar mass. 
In its crown shape, there are 8 Sulphur atoms and we know. 35.967 081(2), [0.000 129, Hint: Sulphur is non-metal belonging to the p-block elements and has the molecular mass as 32.Calculate the molecular weight of sulfur if 35.5 grams of sulfur dissolve in 100.0 grams of CS2 to produce a solution that has a boiling point of 49.48oC.Sulfur S Molar and molecular weight of S S (Sulfur), 1, 32.065, 32.065 Total atoms, Atomic mass, Total mass Proton Number – Atomic Number – Density of Sulfur Note that each element may contain more isotopes. The most abundant (95.02 atom percent) isotope of naturally occurring. Was this answer helpful?Sulfur-32 atom is the stable isotope of sulfur with relative atomic mass 31.972071. 32.07u.Period, 3, Boiling point Block, p, Density (g cm?3) Atomic number, 16, Relative atomic mass State at 20☌, Solid, Key isotopes Electron configuration. 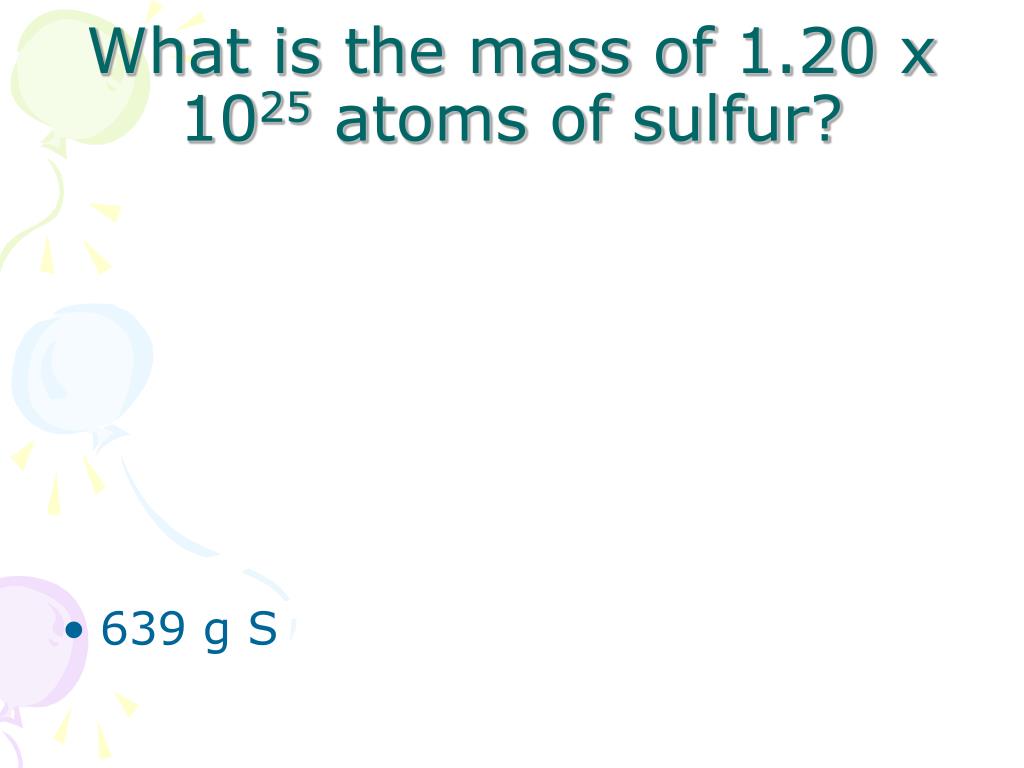
Classified as a nonmetal, Sulfur is a solid at room temperature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
